Description
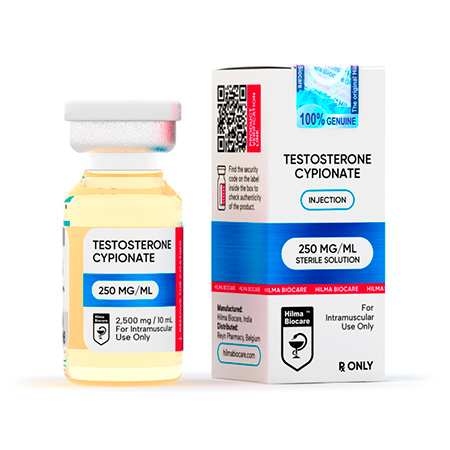
Testosterone Cypionate
Strength: 250 mg/ml
Molecular Formula: C27H40O3,
Molecular Weight: 412.6047 g/mol
Active Ingredient: Testosterone cypionate
CAS number: 58-20-8
Dosage Form: Injectable, oil base sterile solution
Route: Injection
Market Status: Prescription
Company: Hilma Biocare
DESCRIPTION
Testosterone cypionate 250 is an oil based solution for IM injection designed to release
testosterone slowly from the injection site. Testosterone serum concentrations remain
elevated for 7 – 10 days after IM administration. Testosterone cypionate 250 is suitable for
the treatment of hypogonadism and other disorders related to androgen deficiency.
Testosterone cypionate 250 has both anabolic and androgenic effects. Testosterone
supplementation has been demonstrated to increase strength and growth of new muscle
tissue, frequently with increases in libido.
INDICATIONS
Adult Males: Testosterone cypionate 250 injection is indicated for replacement therapy in
conditions associated with a deficiency or absence of endogenous testosterone. Primary
hypogonadism: Testicular failure due to cryptorchidism, bilateral torsion orchitis, vanishing
testis syndrome, or orchidectomy. Hypogonadotropic hypogonadism: Idiopathic
gonadotropin or LHRH deficiency, or pituitary-hypothalamic injury from tumors, trauma, or
radiation.
CLINICAL PHARMACOLOGY
Testosterone and dihydrotestoste- rone are responsible for normal growth and development
of the male sex organs and for maintenance of secondary sex characteristics. These effects
include the growth and maturation of the prostate, seminal vesicles, penis, and scrotum; the
development of male hair distribution, such as facial, pubic, chest, and axillary hair; laryngeal
enlargement; vocal cord thickening: alterations in body musculature; and fat distribution and
have been reported to stimulate the production of red blood cells by enhancing the
production of erythropoietic stimulating factor. Male hypogonadism results from insufficient
secretion of testosterone and is characterized by low serum testosterone concentrations.
Symptoms associated with male hypogonadism include decreased sexual desire with or
without impotence, fatigue and loss of energy, mood depression, regression of secondary
sexual characteristics, and osteoporosis. Hypogonadism is a risk factor for osteoporosis in
men. Androgens have been reported to increase protein anabolism and decrease protein
catabolism. Nitrogen balance is improved only when there is sufficient intake of calories and
protein. During exoger androgens, endogenous testosterone release may be inhibited
through feedback inhibition of pituitary luteinizing hormone (LH). At large doses of
exogenous androgens, spermatogenesis may also be suppressed through feedback
inhibition of pituitary follicle-stimulating hormone (FSH). Esterification of testosterone at
position 17 increases the lipid solubility of the testosterone molecule and prolongs the
activity of the molecule by increasing its residence time. Following intramuscular
administration in an oily vehicle, testosterone ester is slowly absorbed into the circulation
and rapidly hydrolysed in plasma to testosterone. In a study of healthy males, a single
injection of 200 mg of testosterone cypionate increased mean serum testosterone
concentrations sharply to 3 times the basal levels (approximately 1350 ng/dl) at 24 hours
and declined gradually to basal levels (approximately 500 ng/dl) by day 10. Circulating
testosterone is chiefly bound in the serum to sex hormonebinding globulin (SHBG) and
albumin. Testosterone is metabolized to various 17-ketosteroids through two different
pathways. The major active metabolites of testosterone are estradiol and
dihydrotestosterone. Testosterone is metabolized to DHT by steroid 5-alpha reductase
located in the skin, liver, and the urogenital tract of the male. DHT binds with greater affinity
to SHBG than does testosterone.
ADVERSE REACTIONS
Male: Gynecomastia, excessive frequency and duration of penile erections, oligospermia.
Skin and Appendages: Hirsutism, male pattern baldness and acne, gynecomastia.
Fluid/electrolyte Disturbances: Retention of sodium, chloride, water, potassium, calcium, and
inorganic phosphates.
Gastrointestinal: Nausea, cholestatic jaundice, alterations in liver function tests; rarely,
hepatocellular neoplasms, peliosis hepatis, hepatic adenomas, and cholestatic hepatitis.
Hematologic: Suppression of clotting factors II, V, VII, & X; bleeding in patients on anticoagulant therapy.
Nervous System: Increased or decreased libido headache, anxiety, depression, and
generalized paresthesia.
Other: Serum lipid changes, hypercalcaemia, hypertension, oedema, priapism, and
potentiation of sleep apnea.
CONTRAINDICATIONS
Patients with known hypersensitivity to any ingredients in this product. Patients with known
or suspected carcinomas of the breast, testis, or prostate. Patients with severe heart
disease, liver disease, or kidney disease or with a history of epilepsy. Products containing
testosterone should not be used in women as they may cause virilization and fetal harm.
PRECAUTIONS
Because androgens may alter serum cholesterol concentration, caution should be used
when administering these drugs to patients with a history of myocardial infarction or coronary
artery disease Patients on oral anticoagulant therapy require close monitoring especially
when androgens are started or stopped.
Diabetics: androgens may alter the metabolism of oral hypoglycemic agents or may change
insulin sensitivity in patients with diabetes mellitus which may require adjustment of dosage
of insulin and other hypoglycemic drugs.
PATIENT MONITORING
Serum Cholesterol, HDL, LDL, TG. Hemoglobin and Hematocrit, Hepatic function tests –
AST/ALT Prostatic specific antigen – PSA, Testosterone: total, free, and bioavailable.
Dihydrotestosterone & Estradiol. Male patients over 40 should undergo a digital rectal
examination and evaluate PSA prior to androgen use. Periodic evaluations of the prostate
should continue while on androgen therapy, especially in patients with difficulty in urination
or with changes in voiding habits. Use only under the supervision of a qualified physician.
DOSAGE AND ADMINISTRATION
Adult Male: 150 – 300mg injected IM every 1 to 2 weeks or as directed by physician.
PRESENTATION
Testosterone cypionate 250 mg/ml, 10ml multiple dose vial.
STORAGE
Store in a cool dry place between 15 – 25°C. Protectfrom light.







Reviews
There are no reviews yet.